Introduction
The keyword hcooch ch2 h2o refers to a group of fundamental chemical substances that often appear together in discussions of organic synthesis, hydrolysis, and biochemical pathways. These compounds include HCOOCH (commonly known as methyl formate), CH₂ (often interpreted as formaldehyde, CH₂O), and H₂O (water). Each plays a unique role in chemistry, but when combined, they form the basis of important reactions such as ester hydrolysis and organic acid formation.
Studying hcooch ch2 h2o is not just about understanding individual molecules; it highlights the interconnected nature of organic compounds and water in shaping industrial chemistry, pharmaceuticals, and even theories about the origin of life on Earth.
Understanding the Components of HCOOCH CH₂ H₂O
HCOOCH (Methyl Formate)
- Molecular formula: HCOOCH₃
- Family: Ester (derivative of formic acid and methanol)
- Physical properties: Colorless, flammable liquid with a fruity odor
- Uses: Solvent, intermediate in pharmaceuticals, foam-blowing agent
Methyl formate is an ester. Esters are known for their pleasant odors and importance in synthetic chemistry.
CH₂ (Formaldehyde or Methylene Group)
- As CH₂O: Simplest aldehyde, highly reactive
- As CH₂ group: Appears in hydrocarbons and polymer structures
- Importance: Precursor for resins, plastics, and prebiotic compounds
In the context of hcooch ch2 h2o, CH₂ usually refers to formaldehyde (CH₂O), a compound crucial in industrial resin production and biochemical transformations.
H₂O (Water)
- Molecular formula: H₂O
- Role: Universal solvent, hydrolysis agent, stabilizer
- Importance: Enables nearly all biochemical and industrial reactions
Water is the medium and participant in most chemical reactions involving esters and aldehydes.
Also Read: Geometry Spot: A Complete Guide to Interactive Geometry Learning
Key Reactions Involving HCOOCH CH₂ H₂O
Hydrolysis of HCOOCH in Water
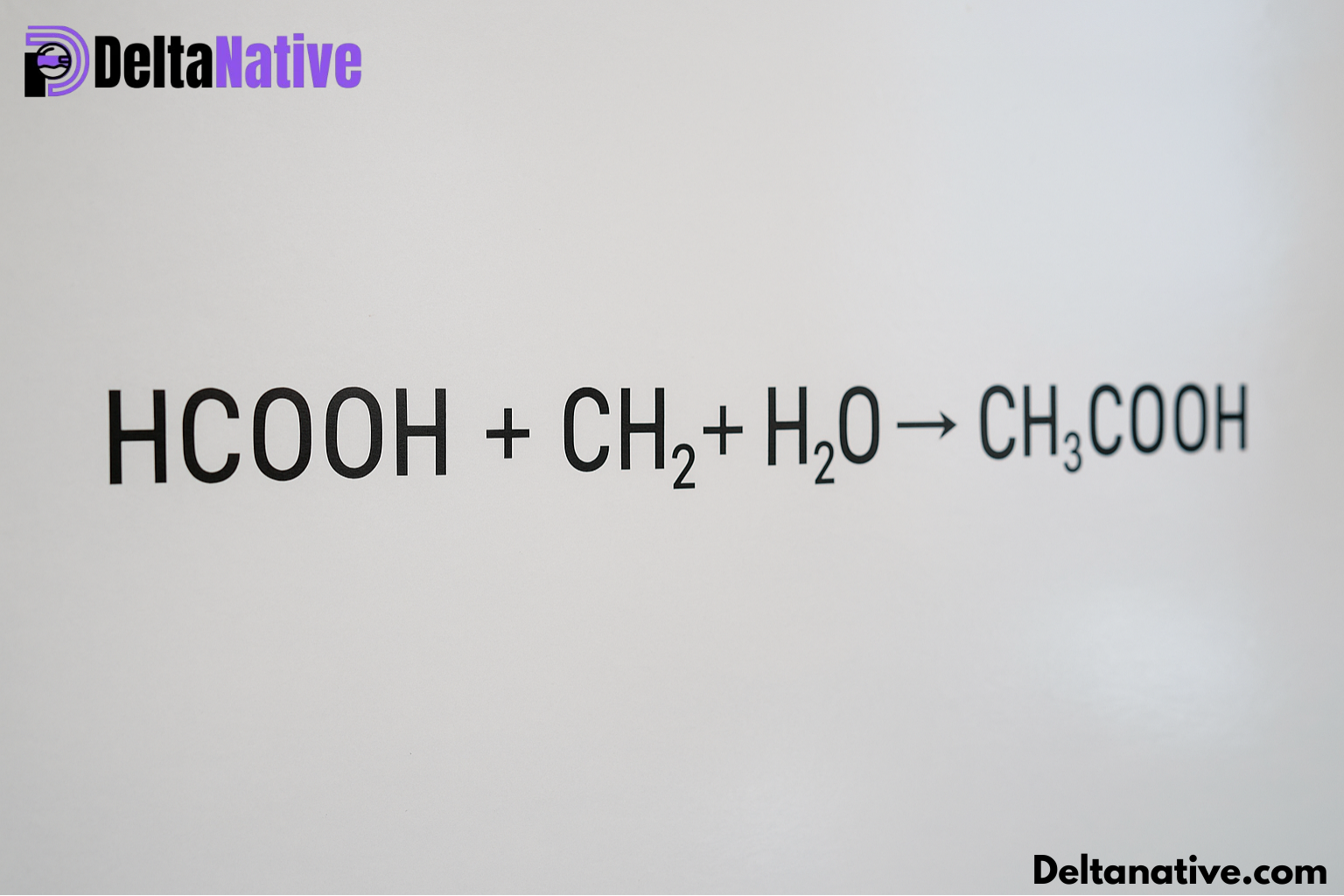
One of the central reactions is ester hydrolysis. HCOOCH3+H2O → HCOOH+CH3OHHCOOCH₃ + H₂O \; \rightarrow \; HCOOH + CH₃OHHCOOCH3+H2O→HCOOH+CH3OH
- Reactants: Methyl formate (HCOOCH₃) + Water (H₂O)
- Products: Formic acid (HCOOH) + Methanol (CH₃OH)
This process can be catalyzed by an acid (HCl) or a base (NaOH). In basic conditions, sodium formate is produced instead of free formic acid.
Reaction with CH₂ (Formaldehyde)
The formic acid (HCOOH) produced from hydrolysis can react with formaldehyde (CH₂O) in water. This leads to the formation of glycolic acid (HOCH₂COOH): HCOOH+CH2O → HOCH2COOHHCOOH + CH₂O \; \rightarrow \; HOCH₂COOHHCOOH+CH2O→HOCH2COOH
This is significant because glycolic acid is used in cosmetics, pharmaceuticals, and polymer chemistry.
Prebiotic Chemistry and Origins of Life
In theories of early Earth chemistry, simple molecules like HCOOH, CH₂O, and H₂O were key in producing more complex organic molecules. The interaction of hcooch ch2 h2o might have been part of pathways that gave rise to amino acids, sugars, and metabolic precursors.
Industrial Applications of HCOOCH CH₂ H₂O
Solvent and Chemical Intermediate
Methyl formate (HCOOCH₃) is widely used as a solvent in:
- Paints
- Coatings
- Adhesives
Resin and Polymer Production
CH₂O (formaldehyde) is an essential raw material in the production of:
- Urea-formaldehyde resins
- Phenol-formaldehyde adhesives
- Melamine resins
These materials are important in wood products, construction, and plastics.
Pharmaceutical and Cosmetic Uses
- Methanol and glycolic acid, derived from these reactions, are used in cosmetics and skin treatments.
- Formic acid finds application in preservatives and antibacterial formulations.
Energy and Fuels
Research suggests methyl formate could be a potential alternative fuel or fuel additive, thanks to its clean-burning properties.
Step-by-Step Guide: Hydrolysis of HCOOCH with H₂O and CH₂
Step 1: Set Up Reaction
- Combine methyl formate (HCOOCH₃) with water (H₂O).
- Add catalyst: either acid (HCl) or base (NaOH).
Step 2: Heat the Mixture
- Gently heat to 40–60°C to accelerate hydrolysis.
Step 3: Hydrolysis Process
- Ester bond breaks down.
- Products: Formic acid + Methanol.
Step 4: Reaction with CH₂
- Add formaldehyde (CH₂O).
- Reaction produces glycolic acid.
Step 5: Product Recovery
- Methanol is separated by distillation.
- Formic acid or glycolic acid collected for industrial use.
Tip: Base-catalyzed hydrolysis is often preferred because it is irreversible and faster.
Scientific Importance of HCOOCH CH₂ H₂O
- Organic Chemistry – Demonstrates ester hydrolysis and aldehyde reactions.
- Biochemistry – Provides insights into small molecule transformations.
- Industrial Chemistry – Foundation for solvents, adhesives, and plastics.
- Prebiotic Studies – Links simple molecules to life’s building blocks.
Also Read: Joincrs com: The Complete Guide for Educators and Students
Conclusion
The study of hcooch ch2 h2o provides a clear example of how simple molecules interact to form essential compounds. Methyl formate (HCOOCH₃) hydrolyzes in water to produce formic acid and methanol, while formaldehyde (CH₂O) reacts further to create glycolic acid.
These reactions are not just laboratory curiosities—they have real industrial uses in resins, solvents, adhesives, and pharmaceuticals, while also offering clues about the chemical origins of life.
In short, hcooch ch2 h2o represents the fascinating intersection of basic organic reactions, industrial applications, and biochemistry.
FAQs About HCOOCH CH₂ H₂O
1. What does hcooch ch2 h2o represent?
It refers to methyl formate (HCOOCH₃), formaldehyde (CH₂O), and water (H₂O).
2. What happens when HCOOCH reacts with water?
It undergoes hydrolysis, producing formic acid and methanol.
3. How does CH₂ (formaldehyde) fit in this reaction?
Formaldehyde reacts with formic acid to form glycolic acid, a valuable compound in chemistry and cosmetics.
4. What are the industrial uses of these compounds?
They are used in adhesives, plastics, solvents, cosmetics, and fuel research.
5. Why is this combination important in chemistry?
It illustrates key concepts like ester hydrolysis, aldehyde reactions, and the role of water as a solvent and reactant.